Introduction: Despite the improved clinical outcomes with novel therapies in patients with chronic lymphocytic leukemia (CLL), Richter transformation (RT) of CLL remains a major clinical challenge. Our group showed a selective response (~40%) with the PD-1 blocking antibody pembrolizumab in patients with RT, particularly after prior exposure to ibrutinib (Ding et al, Blood, 2017). It has been established that PD-1 inhibits cytotoxic T-cell functions via the interaction with its ligands, PD-L1 and PD-L2. However, the role of PD-1 in tumor B-cells is understudied. Here we show the expression and functional implications of the PD-1 signaling axis in B-cell pathobiology.
Methods: CLL-involved lymph node (LN) (n=26) and RT (with diffuse large B-cell lymphoma [DLBCL] histology) -involved LN (n=20) samples were tested for PD-1 expression by immunohistochemistry (IHC) (mouse clone NAT105, Abcam). Then, PD-1 expression was examined in 10 lymphoma cell lines and 1 CLL-like cell line (MEC-1) by both flow cytometry (FC) and Western blot (WB) analysis. PD-1 overexpression using pLEX-lentiviral packaging system (Thermo Scientific) was evaluated for its impact on in vitro cell cycle regulation and in vivo tumor growth. CRISPR/CAS9 mediated PD-1 knockdown evaluated on cell growth in MEC-1 and Mino cells. Role of PD-1 was further examined on the regulation of cell signaling pathways using human phospho kinase array kit (R&D) and WB analysis. Additionally, gene expression signatures were evaluated by RNA-seq using pLEX-lentiviral-PD-1 transfected DLBCL cell lines OCI-LY7 and OCI-LY19.
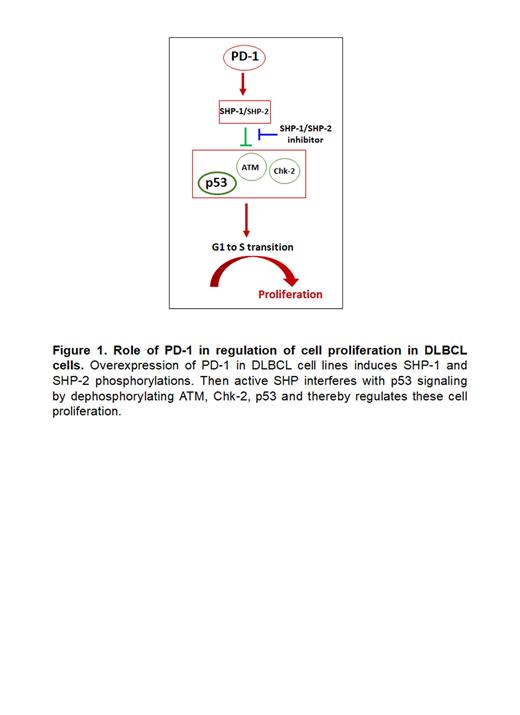
Results: PD-1 expression was significantly increased in RT-LN compared to CLL-LN (mean ± SEM in RT vs. CLL, 30.6% ± 4.7% vs. 11.5% ± 2.8%, p<0.001) as determined by IHC. PD-1 expression was highest in patients with RT where the last prior CLL therapy was ibrutinib. To test the role of PD-1 in tumor B-cells, its expression was assessed in lymphoma and CLL cell lines. The expression of PD-1 was found to be variable by both FC and WB analysis, but at very low-levels in DLBCL cell lines OCI-LY7 and OCI-LY19. Constitutive lentiviral (pLEX-PD-1)-mediated overexpression of PD-1 in OCI-LY7 and OCI-LY19 cells led to increased cell growth (1.4 and 1.9 fold, respectively, compared to original lines after 4 days of culturing of equal number of cells), which was further confirmed by cell cycle analysis which showed an increase in S phase by 20.4% (p<0.05) and 24.5% (p<0.05) in OCI-LY7 and OCI-LY19 cells, respectively. When the luciferase+ PD-1 overexpressing OCI-LY7 cells (1X10 6) were injected intravenously into NSG mice, increased tumor growth (p<0.01) was observed by bioluminescence imaging after 22 days of follow-up. PD-1 knockdown in MEC-1 and Mino cells (high basal PD-1 expression) led to significant inhibition of cell growth (85% and 79.5%, respectively, p<0.05). Using phospho-kinase array, an overall decrease of phosphorylation was detected on multiple sites of p53 (S392, S15, S46) and Chk-2 (T68) in PD-1 overexpressing OCI-LY7 and OCI-LY19 cell lines. This finding was further confirmed by WB analyses. In addition, decreased total p53 and increased phosphorylation on both SHP-1 and SHP-2 were found in PD-1 overexpressing OCI-LY7 and OCI-LY19 cell lines. SHP-1 (TPI-1) and SHP-1+SHP-2 (TPI-1+TNO155) inhibitor treatment promoted OCI-LY19 cell death and led to recovery of phosphorylation on ATM, Chk-2 and p53 in these cells. RNA sequencing data in PD-1 overexpressing OCI-LY7 and OCI-LY19 cells revealed increased expression of genes that promote cell cycle progression and DNA replication.
Conclusion: Our data showed robust PD-1 expression in patients with Richter transformation from CLL to DLBCL. PD-1 overexpression in DLBCL lymphoma cell lines enhanced cell proliferation in vitro and in vivo. Further investigation identified that PD-1 modified the phosphorylation/function of SHP phosphatases and thereby regulated p53 pathways ( Figure 1). These findings indicate that RT with DLBCL cells have the ability to “hijack” the PD-1 pathway which results in downregulation of p53 mediated DNA repair. This novel finding provides for new strategies to further evaluate the interaction of PD-1 signals with DNA repair pathways and suggest potential novel targets for RT.
Disclosures
Sakemura:Janssen Pharmaceutical: Honoraria. He:Kura Oncology, Inc: Consultancy. Wang:Genmab: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Membership on an entity's Board of Directors or advisory committees; Novartis: Research Funding; Genentech: Research Funding; BeiGene: Membership on an entity's Board of Directors or advisory committees; Kite: Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees, Research Funding; Morphosys: Research Funding; LOXO Oncology: Membership on an entity's Board of Directors or advisory committees, Research Funding; Innocare: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Eli Lilly: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy. Klee:Undiganozied Disease Network Int'l: Membership on an entity's Board of Directors or advisory committees; Helix Population Genomics: Patents & Royalties: Helix Population Genomics. Parikh:Boehringer Ingelheim Pharmaceuticals Incc: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Beigene: Membership on an entity's Board of Directors or advisory committees; Genentech: Research Funding; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Vincerx: Research Funding; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Sunesis: Research Funding; CSL Behring: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb-Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Accerta Pharmaceuticals: Research Funding; AbbVie Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding. Shanafelt:Pharmacyclics: Research Funding; AbbVie: Research Funding; Genentech: Research Funding. Ansell:ADC Therapeutics, Affimed, Bristol-Myers Squibb Company, Pfizer Inc, Regeneron Pharmaceuticals Inc, Seagen Inc, Takeda Pharmaceuticals USA Inc.: Other: Contracted Research. Kenderian:Morphosys: Research Funding; Sendero: Patents & Royalties; Torque: Consultancy; Lentigen: Research Funding; Mettaforge: Patents & Royalties; LEAHLabs: Consultancy, Current equity holder in private company, Research Funding; Tolero/Sumtomo: Research Funding; Luminary therapeutics: Other: scientific advisory board ; CapstanBio: Consultancy, Other: Scientific advisory board; Kite/Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Humanigen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding, Speakers Bureau; MustangBio: Patents & Royalties; Novartis: Consultancy, Honoraria, Patents & Royalties, Research Funding, Speakers Bureau; Juno/BMS: Other: Membership on an entity's board of directors or advisory committees, Research Funding. Kay:Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; Vincerx: Research Funding; Sunesis: Research Funding; Beigene: Membership on an entity's Board of Directors or advisory committees; Bristol Meyer Squib / Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; boehringer ingelheim: Membership on an entity's Board of Directors or advisory committees; Pharmcyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Agios Pharm: Membership on an entity's Board of Directors or advisory committees; Genentech: Research Funding; Behring: Membership on an entity's Board of Directors or advisory committees; Acerta Pharma: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees. Ding:Merck: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astra Zeneca: Research Funding; AbbVie: Research Funding; BeiGene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Octapharma: Membership on an entity's Board of Directors or advisory committees, Research Funding; DTRM: Research Funding; MEI Pharma: Membership on an entity's Board of Directors or advisory committees; Alexion: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal